Pembrolizumab-Associated Oral Mucositis: A Global Pharmacovigilance Analysis and External Validation Based on FAERS and EudraVigilance Databases
##plugins.themes.bootstrap3.article.main##
Abstract
Abstract
Background: Pembrolizumab, a programmed cell death protein 1 (PD-1) inhibitor, has fundamentally altered the management of multiple malignancies; however, its immune-related adverse events (irAEs) can compromise treatment outcomes. Among these, oral mucositis (OM) is a clinically significant but often underestimated toxicity. This study aims to characterize the real-world safety signals, temporal dynamics, and risk factors of pembrolizumab-associated OM using global pharmacovigilance databases.
Methods: We performed a retrospective disproportionality analysis using data from the US FDA Adverse Event Reporting System (FAERS) (2014–2025) and the EudraVigilance (EV) database for external validation. Four statistical algorithms—Reporting Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Information Component (IC), and Empirical Bayes Geometric Mean (EBGM)—were utilized for signal detection. Time to onset (TTO), gender-based reporting preferences (log2ROR), and clinical outcome associations were also systematically evaluated.
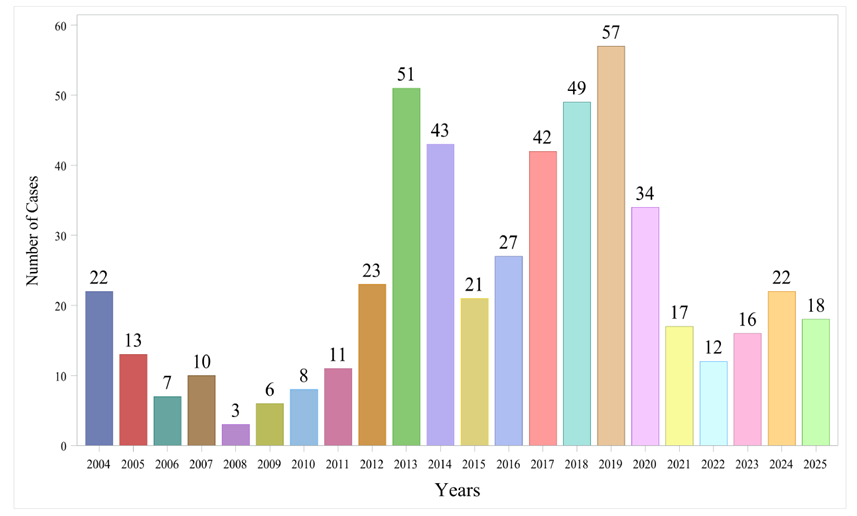
Results: A total of 679 and 816 OM reports were identified in FAERS and EV, respectively. The reporting frequency exhibited a steady upward trajectory, peaking in 2024. In FAERS, OM showed a distinct female predominance (57.58%) and was prevalent in elderly patients (≥65 years, 45.95%). Robust signals were confirmed for "Stomatitis" (ROR 3.78, 95% CI 3.48–4.10) and "Oral mucosa erosion" (ROR 5.06, 95% CI 3.09–8.28), with "Stomatitis haemorrhagic" exhibiting the highest signal intensity (ROR 6.29). The median TTO was 12.50 days (IQR: 4.00–35.00), with 71.74% of events occurring within the first 30 days. Subgroup analysis revealed a significant female susceptibility (EudraVigilance: log2ROR = 0.502, FDR_P < 0.001), while age showed no significant difference after FDR correction. Correlation analysis associated stomatitis primarily with non-fatal clinical outcomes (P < 0.0001).
Conclusion: Pembrolizumab-associated OM is a high-risk, early-onset irAE that manifests predominantly within the first month of therapy. Female and elderly patients are particularly vulnerable, though the toxicity generally follows a non-lethal trajectory. Clinicians should prioritize proactive oral screening and early intervention during the initial 30-day "critical window" to mitigate the high burden of hospitalization and optimize patient quality of life.