TET-1 as Epigenetic Regulator of Ferroptosis in Preeclampsia Pathogenesis
##plugins.themes.bootstrap3.article.main##
Abstract
Background: Preeclampsia, a life-threatening gestational hypertensive syndrome, is pathogenically linked to placental oxidative stress. Emerging evidence implicates ferroptosis in disease progression, though epigenetic regulators of this process remain undefined. The role of Ten-Eleven Translocation 1 (TET-1) in modulating placental redox homeostasis requires systematic investigation.
Methods: We analyzed 126 placental tissues in a retrospective cohort. Multimodal assessments included immunohistochemical quantification of TET-1, HIF1A and SLC7A11 expression, qRT-PCR validation, oxidative stress assays, and bioinformatic interrogation of TET-1 interactomes. Multivariable logistic regression modeled clinical and molecular predictors with age, BMI, TET-1 expression and obstetric history.
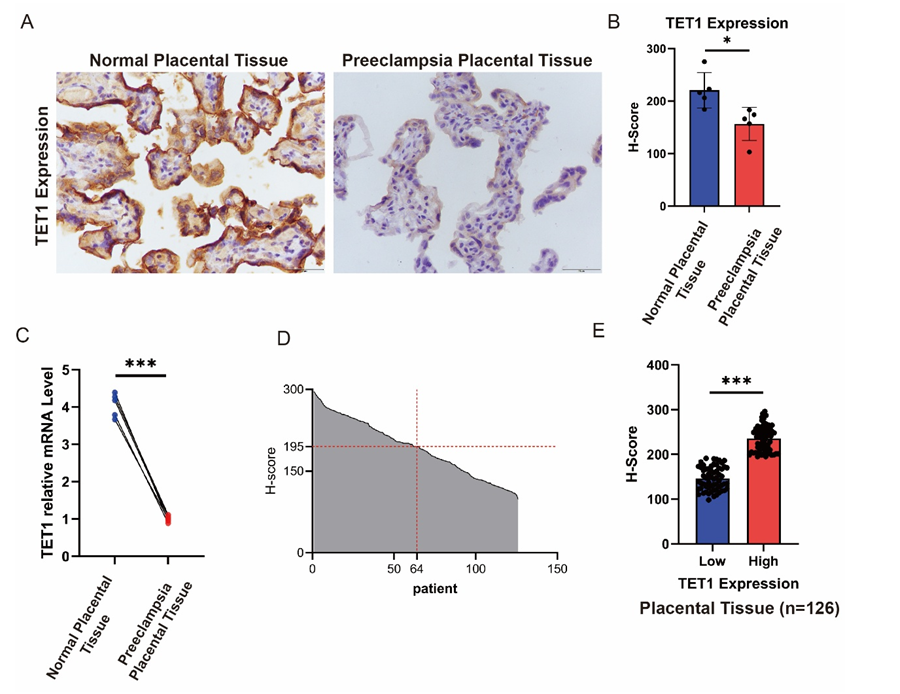
Results: TET-1 expression was significantly suppressed in preeclamptic placentas versus controls (protein IHC: 142.3±18.7 vs 218.9±22.1, P<0.001; mRNA: 0.32±0.11-fold, P<0.001). Reduced TET-1 expression could predict disease risk (adjusted OR=0.172, 95%CI: 0.047–0.638; P=0.008), as did family history (OR=2.952, 95%CI:1.058–8.239; P=0.039). Mechanistically, TET-1 downregulation correlated with elevated HIF-1α methylation and SLC7A11 repression (r=0.489, P=0.0014), forming an epigenetic-ferroptotic axis. Oxidative stress markers increased significantly, with protein interaction validation confirming TET-1- HIF-1α binding.
Conclusion: We discovered TET-1-directed control of the HIF1A-SLC7A11 ferroptosis checkpoint as a potential element of preeclampsia pathogenesis. This epigenetically-gated pathway offers actionable targets for biomarker-driven risk prognostication and molecular therapy in gestational hypertensive disorders.